SOFT MOLECULAR ACTIVATION FOR INTELLIGENT MATERIALS
CATALYZING THE FUTURE OF SMART MATERIALS
-Catalysts developed from soft iodine, graphene and metal complexes could transform smart material technologies.
Research Keywords: Synthetic Chemistry, Catalytic Reaction, Iodine Bond
 ©️Akiko Sato
©️Akiko SatoThe very idea of materials that change their shape or function as required sparks the imagination. Smart materials are impressive artificial molecules, often inspired by naturally occurring chemicals, which have the potential to revolutionize design. They undergo changes that are reversible and repeatable, meaning they can function as molecular switches, valves and sensors. Other smart materials include shape-memory alloys that return to their original shape after bending, metal-bearing polymers that can switch from insulator to conductor and drug-delivery systems that slowly release pharmaceuticals where and when they are needed.
Researchers at the Soft Molecular Activation for Intelligent Materials program are leading investigations into catalysts made from soft molecules that could one day transform smart material design.
Lead researcher Takayoshi Arai says the program is focused on developing powerful catalysts to create novel smart materials using three catalyst types. “Firstly, soft halogen catalysts, predominantly using iodine, can be used to develop highly targeted medications,” he explains. “A second type is ‘soft Π-electron’ using carbon nanotubes and graphene, which would provide a unique reaction sphere for catalysis. The third type, ‘soft metal’ catalysts allow us to introduce soft elements such as gold and copper into target molecules.”
NEW CATALYST DESIGN
Arai says one of the main goals of the program is to create new asymmetric catalysts, which are used to generate large numbers of very specific molecules. Most biological molecules are chiral, meaning they exist in one of two mirror-image forms called enantiomers. Many drugs and smart materials use just one specific enantiomer, and so catalysts are needed to direct the chemical reactions toward forming the correct enantiomer.
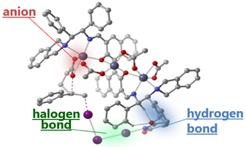
For example, iodine has been used to help synthesize specific enantiomers of cyclic and ring-shaped molecules. “We are lucky that Chiba prefecture in Japan is one of the world’s leading producers of iodine,” says Arai. “In fact, the Chiba Iodine Resource Innovation Center (CIRIC) has opened at Chiba University in July, 2018. A key goal of our program is to perfect iodine-based soft molecular activation.”
Recent success for Arai’s team includes the development of an aminoiminophenoxy-copper carboxylate catalyst that can trigger high yields of O-cyclized molecules. These can, in turn, be used to generate a particular chiral compound that is invaluable for use in antimicrobial and antiosteoporotic compounds.
“A crucial part of our research is designing catalysts that can be reused multiple times, thereby creating cost-effective, sustainable methods for generating useful compounds,” says Arai. His team recently designed a recyclable catalyst made from a zinc-polymeric ligand complex. The polymeric ligand allows the catalyst to be separated from the final product by a simple filtration process, meaning it can be reused — indeed, poly-Zn remained a stable and active catalyst for creating iodine-based ring-shaped molecules more than five times.
“By introducing soft elements to target molecules, our research will facilitate the development of smart materials with smooth and rapid responses,” says Arai. “We welcome anyone interested in research at the forefront of iodine-related soft molecular activation chemistry to come and work in this diverse and challenging field at Chiba.”
(CHIBA RESEARCH 2020)Members
Principal Investigator
| Name | Title, Affiliation | Research Themes |
|---|---|---|
| ARAI Takayoshi | Professor, Graduate School of Science | Organic Chemistry |
Co-Investigatior
| Name | Title, Affiliation | Research Themes |
|---|---|---|
| KANOH Hirofumi | Professor, Graduate School of Science | Colloid Surface Science |
| MATSUMOTO Shoji | Associate Professor, Graduate School of Engineering | Iodine Chemistry |
| MORIYAMA Katsuhiko | Associate Professor, Graduate School of Science | Chemical Reaction |
| YOSHIDA Yasushi | Assistant Professor, Graduate School of Engineering | Catalyst Chemistry |
| ARAI Shigeru | Associate Professor, Graduate School of Pharmaceutical Science | Organic Synthetic Chemistry |
| YOSHIDA Kazuhiro | Associate Professor, Graduate School of Science | Catalyst Chemistry |
| MINO Takashi | Professor, Graduate School of Engineering | Catalyst Chemistry |
| ICHIKUNI Nobuyuki | Professor, Graduate School of Engineering | Catalyst Chemistry |
| OHBA Tomonori | Associate Professor, Graduate School of Science | Catalyst Chemistry |
| YAMADA Yasuhiro | Associate Professor, Graduate School of Engineering | Carbon Material Catalysis |
| KATSUTA Shoichi | Professor, Graduate School of Science | Molecular Spectroscopy, Solution Chemistry |
| SHIROTA Hideaki | Associate Professor, Graduate School of Science | Molecular Spectroscopy, Solution Chemistry |
| MORITA Takeshi | Associate Professor, Graduate School of Science | Physical Chemistry for Molecular Structure |
| MASU Hyuma | Associate Professor, Graduate School of Engineering | Organic Solid Chemistry |
| SAKANE Fumio | Professor, Graduate School of Science | Life Science |
| MURATA Takeshi | Professor, Graduate School of Science | Biostructural Chemistry |
| HASHIMOTO Takuya | Specially Appointed Associate Professor, IGPR/Graduate School of Science | Organic Synthetic Chemistry |
| HARADA Shinji | Assistant Professor, Graduate School of Pharmaceutical Science | Synthetic Organic Chemistry |
| HARADA Shingo | Associate Professor, Graduate School of Pharmaceutical Science | Organic Chemistry |
| UEHARA Tomoya | Professor, Graduate School of Pharmaceutical Science | Radiopharmaceuticals Chemistry |
| TAKADA Mamoru | Assistant Professor, Graduate School of Pharmaceutical Science | Tumor Medical Science |
| NAKAJIMA Masaya | Assistant Professor, Graduate School of Pharmaceutical Science | Organic Chemistry |
| IIDA Keisuke | Assistant Professor, Graduate School of Pharmaceutical Science | Chemical Biology |
| YAMADA Yasuhiro | Associate Professor, Graduate School of Pharmaceutical Science | Ligting Physics |
Research report(2016〜2018)

Following are the representative achievement from our project:
[Soft halogen]
T. Arai developed a halogen-bonding asymmetric catalyst (Chem. Comm. 2018, 54, 3847).
[Soft metal]
T. Arai developed a chiral di-nuclear Pd-catalyst for 1,3-dimine synthesis (Scientific Reports, 2018, 8, 837).
[Soft π-electron and Soft Material]
K. Hoshino developed a metal oxide nanoparticles/viologen composite film electrodes (J. Phys. Chem. C, 2018, 122, 22577).
T. Ohba & Y. Yamada reported CO2 adsorption on Nitrogen-containing Polymer (J. Phys. Chem. C, 2018, 122, 24143).
[Soft Chemical Biology]
M. Ishibashi & M. A. Arai discovered Hedgehog inhibitors (ACS Chem. Biol. 2018, 13, 2551).
Important goal is the integration of individual concepts for soft molecular activations.